2025-07-25 09:51:54
On July 10, 2025, NCCN released the latest update (Version 7.2025) of its Non-Small Cell Lung Cancer (NSCLC) Clinical Practice Guidelines, incorporating Sunvozertinib as a post-line therapy option for advanced or metastatic NSCLC patients with EGFR exon20 insertion mutations (EGFR Exon20ins). This recommendation applies to patients who have progressed after first-line treatment.
This therapeutic regimen was earlier granted accelerated approval by the FDA on July 2, 2025, making it the first and only China-originated EGFR Exon20ins-targeted NSCLC therapy approved in the U.S.

NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Non-Small Cell Lung Cancer (NSCLC)

FDA Approval Letter (July 2, 2025)
The accelerated approval of Sunvozertinib (Zegfrovy®) and its inclusion in clinical guidelines were based on the Phase II WU-KONG1B trial (NCT03974022). This international, multicenter, open-label, dose-randomized study enrolled patients with platinum-refractory, locally advanced/metastatic EGFR Exon20ins NSCLC.
Trial participants were randomly assigned in a 1:1 ratio to two groups: 200 mg/day or 300 mg/day of Sunvozertinib. The efficacy-evaluable population consisted of 85 patients in the 200 mg/day group.
Compared to its FDA approval in July 2025 and inclusion in the NCCN guidelines, Sunvozertinib (Zegfrovy®), a domestically developed EGFR exon20ins NSCLC-targeted therapy, was approved by the NMPA as early as August 2023 and added to the CSCO guidelines in April 2024. Furthermore, it was included in China's National Reimbursement Drug List (NRDL) effective January 1, 2025.

The FDA has accepted Takeda Pharmaceutical's voluntary withdrawal application for mobocertinib (EXKIVITY®)
In the same year, mobocertinib (EXKIVITY®) was withdrawn from the Chinese market
Compared to the two approved drugs (mobocertinib and sunvozertinib), ongoing clinical trials targeting EGFR Exon20ins mutations remain limited. This is primarily due to the low detection rate of Exon20ins mutations—accounting for only 0.3%–2.9% of NSCLC cases in China, even among EGFR-high-prevalence populations—far below the proportion of L858R/19del mutations (which dominate EGFR-driven NSCLC).
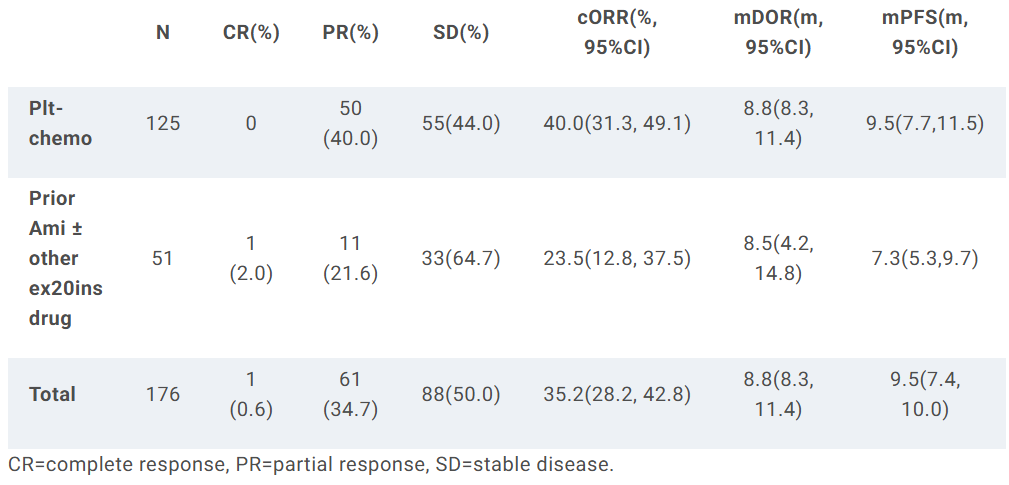
The investigational drug CLN-081 (Zipalertinib), currently in clinical trials, presented Phase 2b data from its REZILIENT1 study at the ASCO meeting in May 2025. The study enrolled 176 patients divided into two cohorts:
- Prior platinum-based chemotherapy alone (n=125)
- Prior platinum-based chemotherapy + amivantamab (n=51)
All patients received 100 mg oral Zipalertinib twice daily.
Key Efficacy Results (median follow-up: 9.3 months):
-
Overall Cohort (n=176):
- Confirmed ORR (cORR): 35.2%
- Median Duration of Response (mDoR): 8.8 months
- Median Progression-Free Survival (mPFS): 9.5 months
-
Subgroup Analysis:
- Naïve to amivantamab (n=125):
- cORR: 40.0%
- mDoR: 8.8 months
- Prior amivantamab exposure (n=51):
- No prior Exon20ins-targeted therapy (n=30):
- cORR: 30.0%
- Prior Exon20ins-targeted therapy (e.g., mobocertinib, sunvozertinib, BLU-451; n=21):
- cORR: 14.3%
- No prior Exon20ins-targeted therapy (n=30):
- Naïve to amivantamab (n=125):
Regulatory Status & Future Potential:
- FDA Breakthrough Therapy Designation (2022) for post-platinum EGFR Exon20ins NSCLC.
- Potential to offer new hope for patients progressing on amivantamab or sunvozertinib.
Source Alignment:
- REZILIENT1 Phase 2b data from ASCO 2025.
- FDA Breakthrough Designation details from 2022.
- Subgroup analysis (amivantamab-experienced) supported by n=51 cohort.
At the 2025 ASCO Annual Meeting, updated clinical data from the REZILIENT1 study (NCT04036682) on CLN-081 (Zipalertinib) were presented, demonstrating its efficacy in EGFR Exon20 insertion (Exon20ins) mutant NSCLC
The following investigational drugs for EGFR Exon20 insertion mutations are currently in clinical development: ABSK112, Poziotinib, and YK-029A. Further robust clinical data are anticipated from these trials.
Compared to the common EGFR L858R and 19del mutations, EGFR Exon20 insertion (Exon20ins) exhibits greater subtype heterogeneity, making it challenging for conventional PCR-based methods to cover all variant types. Thus, next-generation sequencing (NGS) is the clinically preferred detection approach.
1、Detection Methodology:
- NGS outperforms PCR in identifying diverse Exon20ins subtypes due to its high sensitivity and broad coverage.
- PCR limitations: Fails to detect rare/novel Exon20ins variants, potentially missing actionable targets
2、Comprehensive Testing Services:
- Virchow Laboratories offers multi-gene NGS panels covering:
- EGFR mutations (common/rare/Exon20ins).
- Other lung cancer hotspot genes (e.g., ALK, ROS1, MET)
- Applications:Targeted therapy selection (e.g., amivantamab for Exon20ins).Immunotherapy/chemotherapy benefit prediction (e.g., PD-L1/TMB analysis)
The following are selected diagnostic test panels available for clinical use:
-
Lung Cancer Targeted 17-Gene Panel
Sample types: Tissue / Peripheral blood / Pleural/ascitic fluid / Cerebrospinal fluid.
-
Lung Cancer Precision Therapy 107-Gene Panel
Sample types: Tissue / Peripheral blood / Pleural/ascitic fluid / Cerebrospinal fluid.
-
Solid Tumor Targeted 66-Gene Panel
Sample types: Tissue / Peripheral blood / Pleural/ascitic fluid.
-
Solid Tumor Precision Therapy 180-Gene Panel
Sample types: Tissue / Peripheral blood / Pleural/ascitic fluid.
-
Solid Tumor Precision Therapy 715-Gene Panel
Sample types: Tissue / Peripheral blood / Pleural/ascitic fluid
Reference Source:
[1] NCCN非小细胞肺癌指南V7.2025
https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
[2]https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-sunvozertinib-metastatic-non-small-cell-lung-cancer-egfr-exon-20
[3]https://www.federalregister.gov/documents/2024/07/15/2024-15371/takeda-pharmaceuticals-usa-inc-withdrawal-of-approval-of-new-drug-application-for-exkivity
[4]https://www.takeda.com.cn/statements/2024/announcement-for-exkivity-in-china/
[5]Burnett H, et al. Epidemiological and clinical burden of EGFR Exon 20 insertion in advanced non-small cell lung cancer: A systematic literature review. PLoS One. 2021 Mar 8;16(3):e0247620. doi: 10.1371/journal.pone.0247620
[6]https://meetings.asco.org/abstracts-presentations/247323




