丨 About Molecular Pathology
The NGS platform combines pathological and PCR detection analysis methods to examine clinically common solid tumor-related genes. The scope includes single nucleotide variations (SNVs), insertions and deletions (InDels), copy number variations (CNVs), and fusions (Fusions). The analysis covers sensitivity and resistance predictions for targeted, immune, and chemotherapy drugs, as well as genetic risk assessment, prognosis evaluation, molecular subtyping, and recurrence monitoring. This provides patients with more treatment possibilities and assists doctors in effectively guiding and dynamically monitoring the full-cycle treatment of cancer patients.
丨 Testing Content
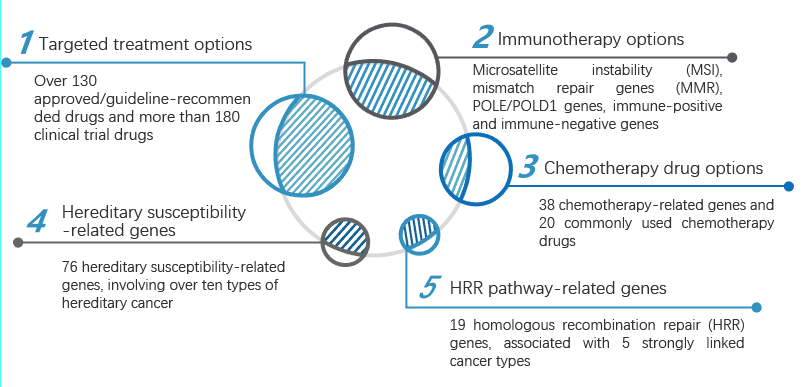
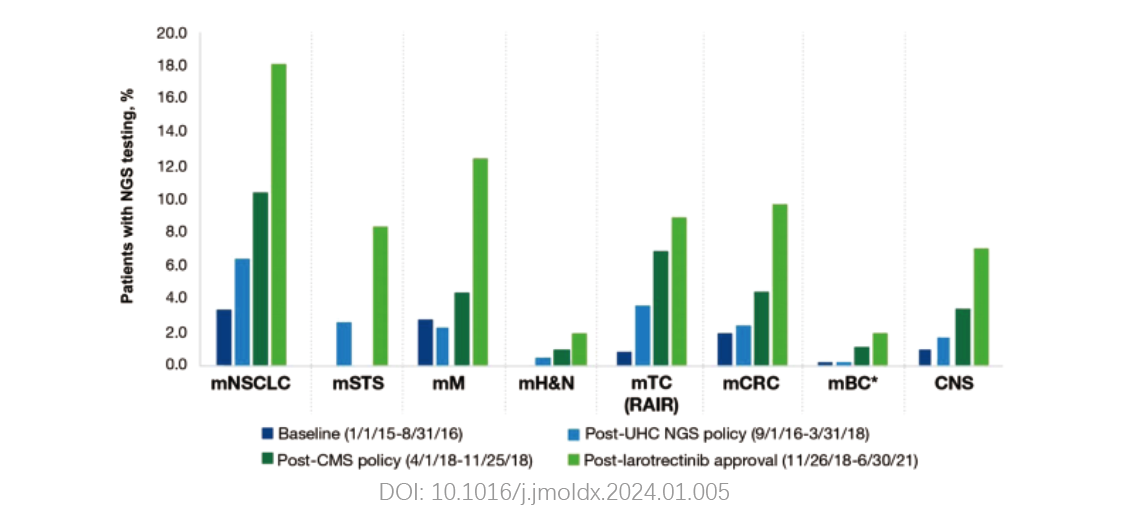
The adoption rate of next-generation sequencing (NGS) among advanced solid tumor patients has been increasing annually, with NGS testing now integrated into over 70% of tumor patient management workflows.
丨 Clinical significance of testing
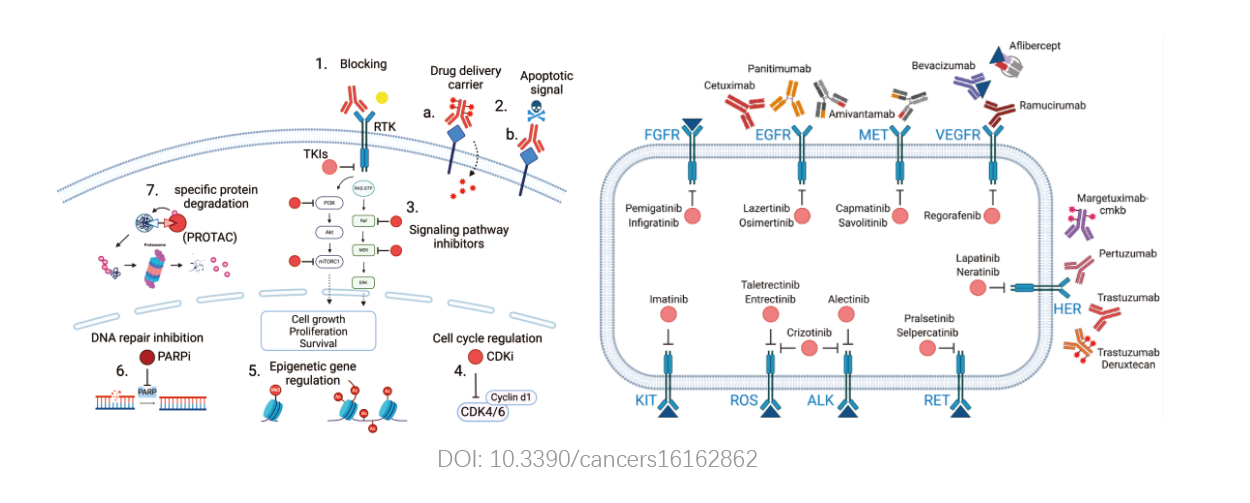
The product's testing scope covers genes and targets related to drugs approved by the U.S. Food and Drug Administration (FDA), China's National Medical Products Administration (NMPA), and major clinical guidelines. Its comprehensive and regularly updated database provides reference information for clinical frontline targeted treatment strategies.
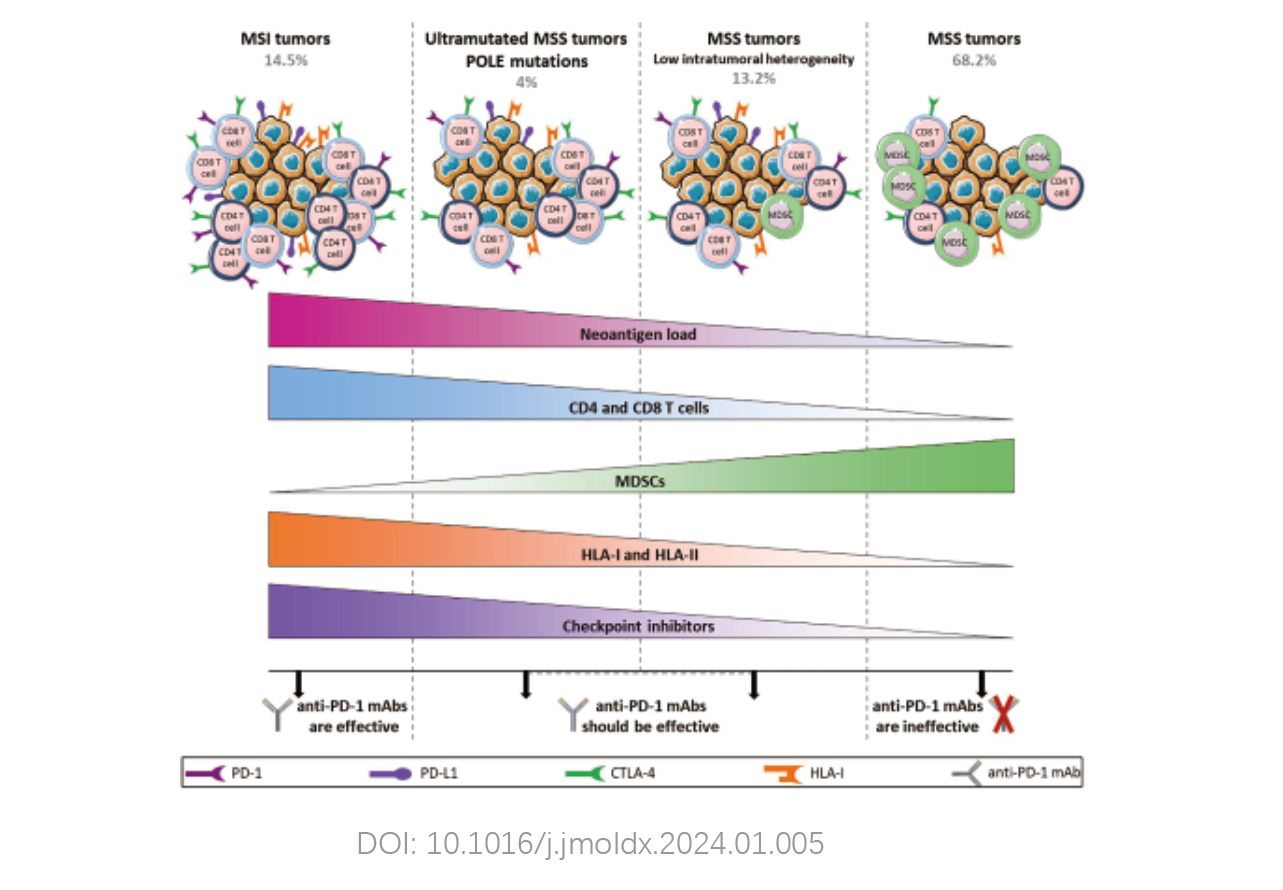
The test includes multiple indicators such as microsatellite instability (MSI), DNA mismatch repair genes (MMR), POLE/POLD1 genes, as well as immune therapy-positive, negative, and hyperprogression-related genes. It predicts patient response to immunotherapy, providing additional information and possibilities for clinical decision-making in immunotherapy.
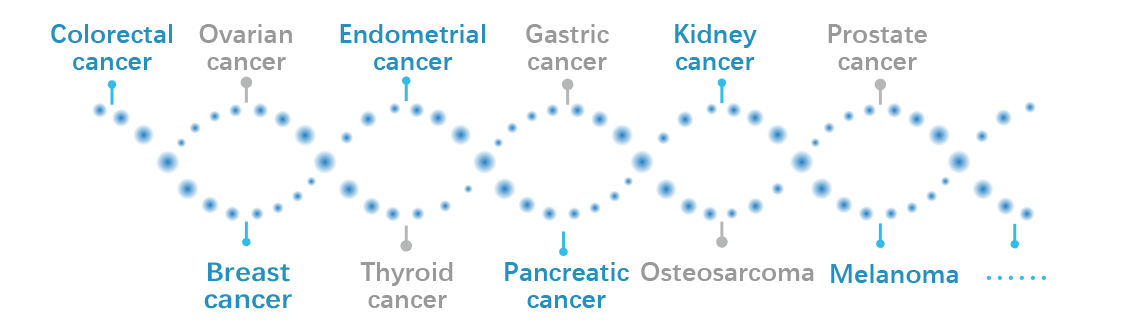
The test covers over 70 genes associated with more than ten types of hereditary cancer, enabling genetic counseling and family screening to facilitate early detection and prevention, thereby reducing the risks of germline inheritance.
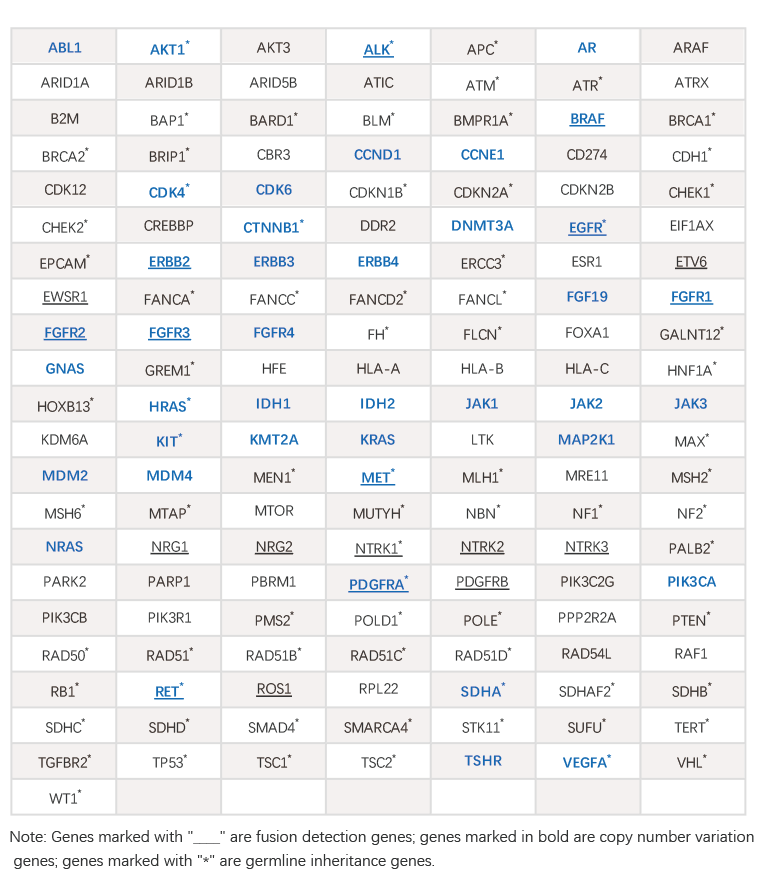
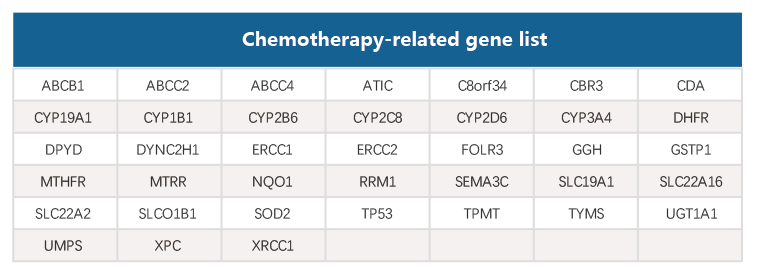
丨 Gene List
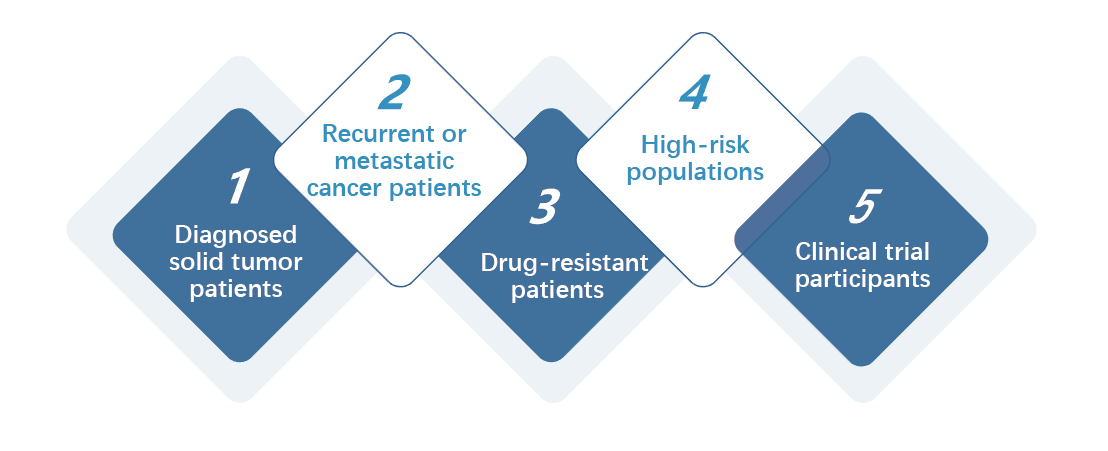
丨 Applicable population
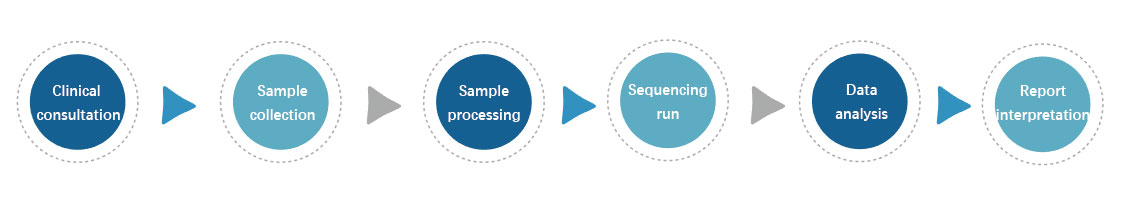
丨 Testing process
杭州魏尔啸实验室
丨 About Molecular Pathology
The NGS platform combines pathological and PCR detection analysis methods to examine clinically common solid tumor-related genes. The scope includes single nucleotide variations (SNVs), insertions and deletions (InDels), copy number variations (CNVs), and fusions (Fusions). The analysis covers sensitivity and resistance predictions for targeted, immune, and chemotherapy drugs, as well as genetic risk assessment, prognosis evaluation, molecular subtyping, and recurrence monitoring. This provides patients with more treatment possibilities and assists doctors in effectively guiding and dynamically monitoring the full-cycle treatment of cancer patients.
丨 Testing Content


The adoption rate of next-generation sequencing (NGS) among advanced solid tumor patients has been increasing annually, with NGS testing now integrated into over 70% of tumor patient management workflows.
丨 Clinical significance of testing

The product's testing scope covers genes and targets related to drugs approved by the U.S. Food and Drug Administration (FDA), China's National Medical Products Administration (NMPA), and major clinical guidelines. Its comprehensive and regularly updated database provides reference information for clinical frontline targeted treatment strategies.

The test includes multiple indicators such as microsatellite instability (MSI), DNA mismatch repair genes (MMR), POLE/POLD1 genes, as well as immune therapy-positive, negative, and hyperprogression-related genes. It predicts patient response to immunotherapy, providing additional information and possibilities for clinical decision-making in immunotherapy.

The test covers over 70 genes associated with more than ten types of hereditary cancer, enabling genetic counseling and family screening to facilitate early detection and prevention, thereby reducing the risks of germline inheritance.
丨 Gene List


丨 Applicable population

丨 Testing process
