一、Projects Offered
1. Paraffin Section Diagnosis
2. Frozen Section Diagnosis
二、Instructions for Submission and Collection Requirements
1. Application Form Completion
Fill out the corresponding special application form of our center according to the testing requirements. Patient information is crucial for pathological diagnosis. Clinicians should carefully and detailedly fill in all items on the application form (including the doctor's name and contact number). In particular, meaningful positive signs, imaging examinations, and other test results should be filled in. If pathological examinations (including cytology and histology) have been previously conducted, the results should be filled in. For those examined in our center, the corresponding pathological number should be filled in. For gynecological specimens, menstrual and pregnancy history should be filled in. The handwriting should be clear and legible.
2. Sample Acquisition
1 ) Tissue Sample Acquisition
① For pathological diagnosis, the more tissue, the higher the diagnostic success rate. Therefore, clinicians should try to obtain as much tissue as possible when excising the tissue, provided that the patient's condition allows.
② When obtaining the tissue, try to obtain non-necrotic tissue. The central area of larger tumors often necroses, so avoid the necrotic area when performing punctures.
③ The tissue-taking action should be gentle, and squeezing of the tissue should be avoided as much as possible. The tissue-taking knife should be sharp, and care should be taken not to use toothed forceps or clamps for obtaining. Mechanical compression or finger kneading should be avoided to prevent artificial deformation, which may hinder diagnosis.
④ Tumor resection specimens should be marked with their orientation, and clearly recorded in the application form, so that the status of each resection margin can be reported when issuing the report. It is recommended to mark the orientation of the following specimens.
2 ) Tissue Specimen Fixation
① The specimen should be submitted intact. Avoid dividing the same specimen for submission to different institutions. If cutting is necessary, ensure all sections are made in the same orientation1.
② Specimens must be fixed promptly. Unless otherwise specified, use 10% neutral buffered formalin for fixation25.
③ The fixative-to-specimen volume ratio should exceed 10:1. For larger specimens, ensure complete immersion in the fixative if the ratio cannot be met5.
④ Use standardized specimen containers provided by the company. The container opening should be wide enough to allow easy placement and retrieval without compression or forced insertion1.
⑤ Clearly label the container with the patient's name, specimen type, anatomical site, and number of tissue blocks.
丨Immunohistochemistry (IHC)
一、Projects Offered
1. Special Staining
2. Fully Automated Immunohistochemical Staining
二、Test Items
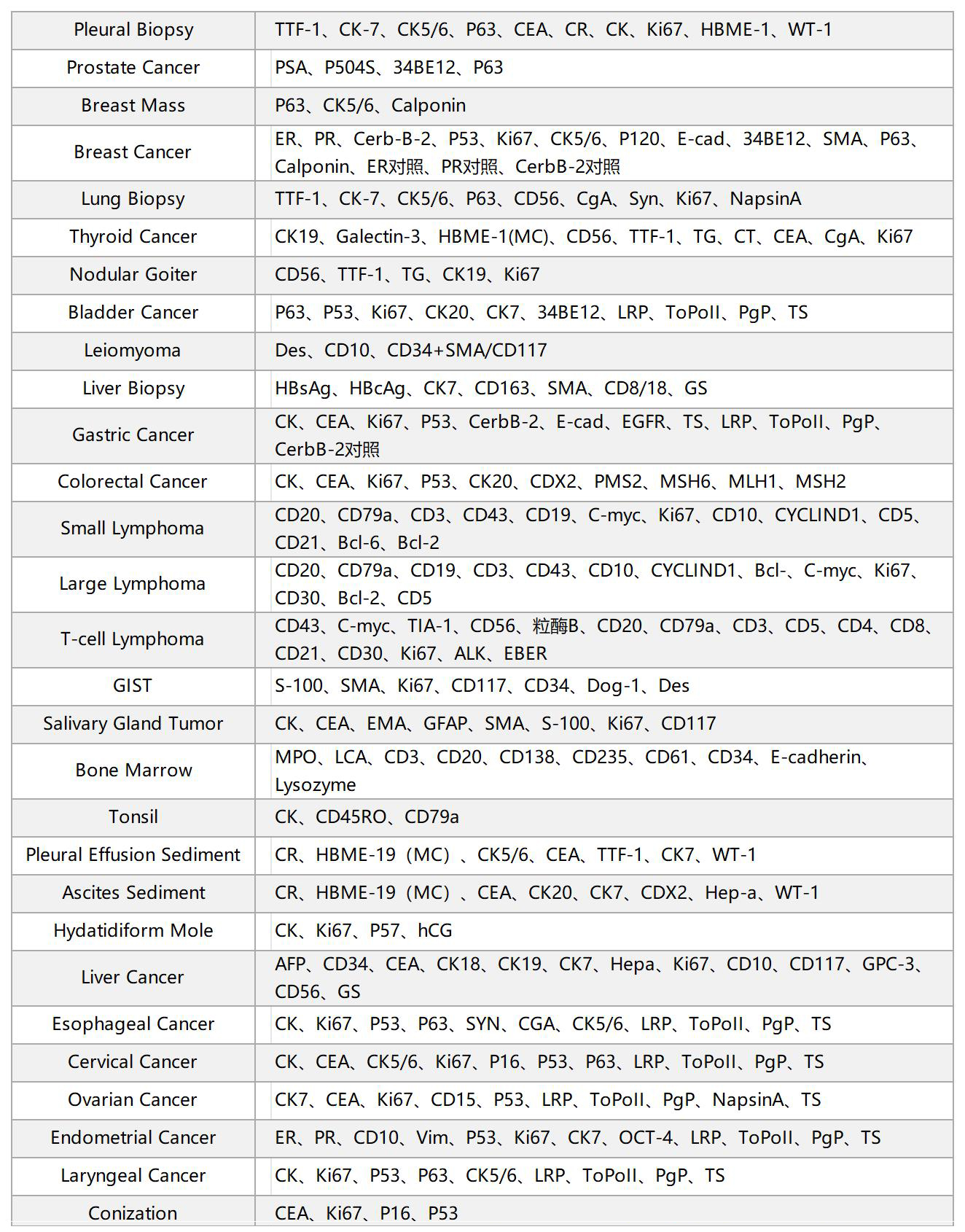
三、Instructions for Submission and Collection Requirements
1. Surgical specimens:
① Fix the specimen with 10% neutral buffered formalin as soon as possible (within 30 minutes) after excision.
② The volume of formalin should be more than 10 times the volume of the tumor tissue.
③ The fixation time should be 6 to 24 hours for surgical specimens and 4 to 12 hours for biopsy specimens.
2. Surgical specimens:
① Fix the specimen with 10% neutral buffered formalin as soon as possible (within 30 minutes) after excision.
② The volume of formalin should be more than 10 times the volume of the tumor tissue.
③ The fixation time should be 6 to 24 hours for surgical specimens and 4 to 12 hours for biopsy specimens.
丨 Cytopathology
一、Projects Offered
1. Gynecological cytology:conventional cytology smear; thin-layer liquid-based cytology (TCT/LCT);
2. non-gynecological cytology:non-gynecological smears (including sputum, urine, pleural and peritoneal effusion, bronchial brushings, etc.);
3. fine needle aspiration cytology (FNAC).
二、Instructions for Submission and Collection Requirements
1. Cervical smear
(1) Abstain from sexual activity for 48 hours before the examination, and avoid vaginal douching and medication.
(2) Avoid the menstrual period.
(3) Fully expose the cervix, and using the external cervical os as the center, gently scrape or brush the squamocolumnar junction and the cervical canal for 1-2 weeks with a special scraper or cervical brush.
(4) For those with irregular vaginal bleeding, use a sterile cotton swab to gently wipe away the blood before obtaining the sample.
(5) For those with excessive vaginal discharge, use a sterile cotton swab to gently wipe away the mucus before obtaining the sample.
(6) Immediately spread the scraped cells onto a clean slide in one motion, smoothly and evenly pushing them towards the right 2/3 of the slide in the same direction. Avoid back-and-forth smearing.
(7) Clearly write the patient's name/ID on the frosted end of the slide.
(8) Immediately fix the smear in 95% alcohol for at least 15 minutes.
(9) Remove the slide from the 95% alcohol, let it air dry, and then place the cell slides in order in a self-sealing bag or slide box, depending on the number of slices or as agreed upon with the pathology laboratory.
2. Liquid-Based Cytology (LBC)
(1)Before the Examination:Avoid sexual intercourse, vaginal douching, and medication use 48 hours prior to the test.
(2)Menstruation:The test should not be performed during menstruation.
(3)Sample Collection:Fully expose the cervix.Gently insert the central bristles of the broom-shaped sampler into the cervical os.Rotate the sampler 5–10 times in the same direction (do not rotate back and forth).
(4)For Patients with Irregular Vaginal Bleeding:Gently wipe away the blood with a sterile cotton swab before sampling.
(5)For Patients with Excessive Vaginal Discharge:Gently remove excess mucus with a sterile cotton swab before sampling.
(6)Sample Preservation:Place the broom-shaped sampler into the preservative solution bottle.
Rinse the sampler by pushing it to the bottom of the bottle 10 times, ensuring the bristles fully disperse.Finally, swirl the sampler rapidly in the solution to further release cells.Securely tighten the bottle cap.
(7)Labeling:Clearly write the patient’s name and age on the specimen bottle label.Verify the information matches the requisition form.
3. Pleural effusion、 Ascites
Handling Instructions for Bloody Pleural Effusion/Ascites:
(1)Anticoagulation Requirement:Heparin anticoagulation should be used for bloody pleural effusion or ascites.
(2)Sample Submission & Processing:Submit as much fluid as possible, and prepare slides promptly.
4. Sputum
(1)Optimal Sample Selection:Morning's first sputum is preferable but not mandatory; blood-tinged sputum is ideal.
(2)Specimen Quality Control:Ensure the sample is from deep coughing (lower respiratory tract), not saliva or oropharyngeal secretions.
(3)Container & Preservation:Use sterile, airtight containers to prevent desiccation during transport.
(4)Post-Bronchoscopy Samples:The first sputum after bronchoscopy shows higher pathogen detection rates and should be submitted.
(5)Sampling Frequency:Collect at least three consecutive specimens for improved diagnostic yield.
(6)Timely Processing:Deliver samples immediately (<2 hours) to the lab to prevent cellular degeneration or bacterial overgrowth.
5. Urine Test
(1)Optimal Sample Timing:The first morning urine is preferred but not mandatory; hematuria (blood in urine) can be submitted at any time.
(2)Sample Volume:Submit the entire voided urine specimen; larger volumes may improve detection rates.
(3)Post-Cystoscopy Samples:The first urine after cystoscopy has a higher diagnostic yield and should be collected.
(4)Sampling Frequency:Collect specimens for at least three consecutive days.
(5)Timely Processing:Deliver samples promptly to the lab to prevent cellular degeneration (ideally within 2 hours).
杭州魏尔啸实验室
一、Projects Offered
1. Paraffin Section Diagnosis
2. Frozen Section Diagnosis
二、Instructions for Submission and Collection Requirements
1. Application Form Completion
Fill out the corresponding special application form of our center according to the testing requirements. Patient information is crucial for pathological diagnosis. Clinicians should carefully and detailedly fill in all items on the application form (including the doctor's name and contact number). In particular, meaningful positive signs, imaging examinations, and other test results should be filled in. If pathological examinations (including cytology and histology) have been previously conducted, the results should be filled in. For those examined in our center, the corresponding pathological number should be filled in. For gynecological specimens, menstrual and pregnancy history should be filled in. The handwriting should be clear and legible.
2. Sample Acquisition
1 ) Tissue Sample Acquisition
① For pathological diagnosis, the more tissue, the higher the diagnostic success rate. Therefore, clinicians should try to obtain as much tissue as possible when excising the tissue, provided that the patient's condition allows.
② When obtaining the tissue, try to obtain non-necrotic tissue. The central area of larger tumors often necroses, so avoid the necrotic area when performing punctures.
③ The tissue-taking action should be gentle, and squeezing of the tissue should be avoided as much as possible. The tissue-taking knife should be sharp, and care should be taken not to use toothed forceps or clamps for obtaining. Mechanical compression or finger kneading should be avoided to prevent artificial deformation, which may hinder diagnosis.
④ Tumor resection specimens should be marked with their orientation, and clearly recorded in the application form, so that the status of each resection margin can be reported when issuing the report. It is recommended to mark the orientation of the following specimens.
2 ) Tissue Specimen Fixation
① The specimen should be submitted intact. Avoid dividing the same specimen for submission to different institutions. If cutting is necessary, ensure all sections are made in the same orientation1.
② Specimens must be fixed promptly. Unless otherwise specified, use 10% neutral buffered formalin for fixation25.
③ The fixative-to-specimen volume ratio should exceed 10:1. For larger specimens, ensure complete immersion in the fixative if the ratio cannot be met5.
④ Use standardized specimen containers provided by the company. The container opening should be wide enough to allow easy placement and retrieval without compression or forced insertion1.
⑤ Clearly label the container with the patient's name, specimen type, anatomical site, and number of tissue blocks.
丨Immunohistochemistry (IHC)
一、Projects Offered
1. Special Staining
2. Fully Automated Immunohistochemical Staining
二、Test Items

三、Instructions for Submission and Collection Requirements
1. Surgical specimens:
① Fix the specimen with 10% neutral buffered formalin as soon as possible (within 30 minutes) after excision.
② The volume of formalin should be more than 10 times the volume of the tumor tissue.
③ The fixation time should be 6 to 24 hours for surgical specimens and 4 to 12 hours for biopsy specimens.
2. Surgical specimens:
① Fix the specimen with 10% neutral buffered formalin as soon as possible (within 30 minutes) after excision.
② The volume of formalin should be more than 10 times the volume of the tumor tissue.
③ The fixation time should be 6 to 24 hours for surgical specimens and 4 to 12 hours for biopsy specimens.
丨 Cytopathology
一、Projects Offered
1. Gynecological cytology:conventional cytology smear; thin-layer liquid-based cytology (TCT/LCT);
2. non-gynecological cytology:non-gynecological smears (including sputum, urine, pleural and peritoneal effusion, bronchial brushings, etc.);
3. fine needle aspiration cytology (FNAC).
二、Instructions for Submission and Collection Requirements
1. Cervical smear
(1) Abstain from sexual activity for 48 hours before the examination, and avoid vaginal douching and medication.
(2) Avoid the menstrual period.
(3) Fully expose the cervix, and using the external cervical os as the center, gently scrape or brush the squamocolumnar junction and the cervical canal for 1-2 weeks with a special scraper or cervical brush.
(4) For those with irregular vaginal bleeding, use a sterile cotton swab to gently wipe away the blood before obtaining the sample.
(5) For those with excessive vaginal discharge, use a sterile cotton swab to gently wipe away the mucus before obtaining the sample.
(6) Immediately spread the scraped cells onto a clean slide in one motion, smoothly and evenly pushing them towards the right 2/3 of the slide in the same direction. Avoid back-and-forth smearing.
(7) Clearly write the patient's name/ID on the frosted end of the slide.
(8) Immediately fix the smear in 95% alcohol for at least 15 minutes.
(9) Remove the slide from the 95% alcohol, let it air dry, and then place the cell slides in order in a self-sealing bag or slide box, depending on the number of slices or as agreed upon with the pathology laboratory.
2. Liquid-Based Cytology (LBC)
(1)Before the Examination:Avoid sexual intercourse, vaginal douching, and medication use 48 hours prior to the test.
(2)Menstruation:The test should not be performed during menstruation.
(3)Sample Collection:Fully expose the cervix.Gently insert the central bristles of the broom-shaped sampler into the cervical os.Rotate the sampler 5–10 times in the same direction (do not rotate back and forth).
(4)For Patients with Irregular Vaginal Bleeding:Gently wipe away the blood with a sterile cotton swab before sampling.
(5)For Patients with Excessive Vaginal Discharge:Gently remove excess mucus with a sterile cotton swab before sampling.
(6)Sample Preservation:Place the broom-shaped sampler into the preservative solution bottle.
Rinse the sampler by pushing it to the bottom of the bottle 10 times, ensuring the bristles fully disperse.Finally, swirl the sampler rapidly in the solution to further release cells.Securely tighten the bottle cap.
(7)Labeling:Clearly write the patient’s name and age on the specimen bottle label.Verify the information matches the requisition form.
3. Pleural effusion、 Ascites
Handling Instructions for Bloody Pleural Effusion/Ascites:
(1)Anticoagulation Requirement:Heparin anticoagulation should be used for bloody pleural effusion or ascites.
(2)Sample Submission & Processing:Submit as much fluid as possible, and prepare slides promptly.
4. Sputum
(1)Optimal Sample Selection:Morning's first sputum is preferable but not mandatory; blood-tinged sputum is ideal.
(2)Specimen Quality Control:Ensure the sample is from deep coughing (lower respiratory tract), not saliva or oropharyngeal secretions.
(3)Container & Preservation:Use sterile, airtight containers to prevent desiccation during transport.
(4)Post-Bronchoscopy Samples:The first sputum after bronchoscopy shows higher pathogen detection rates and should be submitted.
(5)Sampling Frequency:Collect at least three consecutive specimens for improved diagnostic yield.
(6)Timely Processing:Deliver samples immediately (<2 hours) to the lab to prevent cellular degeneration or bacterial overgrowth.
5. Urine Test
(1)Optimal Sample Timing:The first morning urine is preferred but not mandatory; hematuria (blood in urine) can be submitted at any time.
(2)Sample Volume:Submit the entire voided urine specimen; larger volumes may improve detection rates.
(3)Post-Cystoscopy Samples:The first urine after cystoscopy has a higher diagnostic yield and should be collected.
(4)Sampling Frequency:Collect specimens for at least three consecutive days.
(5)Timely Processing:Deliver samples promptly to the lab to prevent cellular degeneration (ideally within 2 hours).